Classification
(and lichenization & de-lichenization)
Humans have a penchant for classifying both living and non-living objects. Objects that are similar in some way can be grouped and given a name that allows them to be referred to as a group. An example of a group name is Homo sapiens and another example is Homo habilis. Technically these are examples of species names. The fact that both species names have Homo as the first element signifies that there is strong enough similarity between the members of the species Homo sapiens and those of the species Home habilis to warrant combining those two species into a larger group, a genus with the group name Homo. So far you have seen an example of individual organisms grouped together into a species and an example of species grouped into a genus. The process does not stop there since groups of similar genera are grouped into a family and, going to still broader groupings (or higher levels of classification), there are orders, classes, phyla and finally kingdoms. This process yields a hierarchical arrangement of groupings and such a hierarchy is an example of a classification scheme. This particular classification system is often referred to as the Linnean classification, though the use of some of the elements predates Linnaeus.
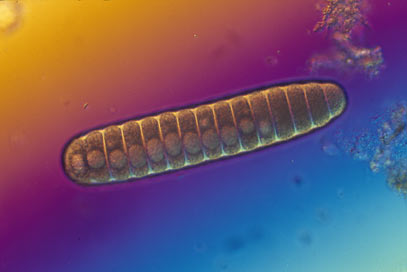
I have emphasized that there is some notion of similarity behind any classification scheme but I have not yet said how similarity is defined. That is a critical point which will be the subject of the next section. For the moment I will assume that there are definitions of similarity for lichens and plants and I will finish this section with some examples of classifications and a few more definitions. For the purpose of illustration here are classifications of the lichen Pannaria sphinctrina ![]() , Acacia pycnantha ( Australia's national flower, the Golden Wattle) and the grass Themeda triandra (Kangaroo Grass) .
, Acacia pycnantha ( Australia's national flower, the Golden Wattle) and the grass Themeda triandra (Kangaroo Grass) .
kingdom: |
Fungi |
Plantae |
Plantae |
phylum: |
Ascomycota |
Magnoliophyta |
Magnoliophyta |
class: |
Lecanoromycetes |
Magnoliopsida |
Liliopsida |
order: |
Lecanorales |
Fabales |
Poales |
family: |
Pannariaceae |
Mimosaceae |
Poaceae |
genus: |
Pannaria |
Acacia |
Themeda |
species: |
Pannaria sphinctrina |
Acacia pycnantha |
Themeda triandra |
'Species' is an example of a taxonomic rank while Pannaria sphinctrina, a particular species, is an example of a taxon. Moreover, it is a taxon at species rank. Genus, family, order, class, phylum and kingdom are also examples of taxonomic ranks. Lecanorales, Physciaceae, Fabales, Poaceae, Acacia and Themeda triandra are more examples of taxa, each being a taxon at some rank. In this hierarchical ordering of living organisms, the total number of species is greater than the number of genera, which in turn is greater than the number of families and so on. There are rules governing the creation of taxonomic names and one is that when a biologist proposes the creation of a new taxon at some taxonomic rank, he or she must ensure that there is at least one new taxon at each lower taxonomic rank. For example, a botanist who wishes to define a new order must ensure that within the new order there is at least one family, which in turn must contain at least one genus, which in turn must contain at least one species.
Warning: If you're looking for the full classification hierarchy for every lichen, this page is not for you. Nor will this page help you identify lichen specimens. Rather, the intent here is to give you some examples of the features or tools used in lichen identification and some general observations about lichen classification. Lichens are often referred to as lichenized fungi and it is the fungal partner on which the classification of a lichen is based. Therefore lichen classification is part of the subject of fungal classification. Moreover, since almost all lichens are lichenized ascomycetes the subject of lichen classification is almost totally part of the topic of ascomycete classification and you can find current thoughts about ascomycete classification on the Myconet website: http://www.fieldmuseum.org/myconet/. On that page click on current outline for the latest classification scheme, down to generic level. You'll find identification guides for Australian lichens listed on the FURTHER READING page. |
Features used in lichen classification
At any given time, anyone attempting to classify lichens can use only those features that the available technology reveals. Several hundred years ago all that was available was the naked eye so features such as growth form, colour and substrate were used to differentiate lichens and by 1700 the concepts of genus and species were in use. Since the 1700s the development of tools or techniques such as optical microscopes, chemical testing, growth studies, electron microscopes and DNA analysis have revealed details about aspects such as lichen structure, physiology, metabolic products, ecology and genetics. Each new tool or method has provided information previously unobtainable. Sometimes this is simply an improvement in seeing something previously known. Early optical microscopes allowed the detection of spores, but with no fine detail of spore appearance, whereas today's optical microscopes reveal finer spore detail. Spores vary in shape, colour, size and septation depending on species. In other cases the new information is astounding and can generate considerable controversy. Until well into the 1800s lichens had been thought of as single organisms. With the help of the optical microscope Simon Schwendener was able to reveal the dual nature of lichens in a paper published in 1867, yet this idea was vehemently attacked by some influential lichenologists for some decades.
Whenever new information has become available it has inevitably supported some existing ideas of lichen classification but contradicted others. As long as new information is capable of being found lichen classification is based on unavoidably incomplete information and so classification schemes are 'works in progress'. A newly found contradiction simply means that some earlier concepts of taxon demarcation need to be re-examined since all the available evidence should be assessed. I have said that early classification was based on naked eye features. In the 19th century microscopic examination showed that some seemingly quite different species were very similar microscopically. Studies in the field showed there to be good evidence that at times the environment could influence thallus morphology, whereas microscopic features remained constant.
The example of STICTA AND 'DENDRISCOCAULON' shows also that thallus morphology could depend on whether the mycobiont was associated with an alga or a cyanobacterium. This meant that heavy reliance on macroscopic morphology as the basis for a classification scheme was untenable since no scientific classification should be based on features that were not fixed, but could vary depending on where a thallus grew or which photobiont occurred in the thallus. By the end of the 19th century increased weight was given to microscopic features in the definition of taxa, though macroscopic morphology was not ignored. One result of this work was that some species, once thought to be distinct, were shown to be identical. Such groups of species were lumped into one. Conversely, there were also cases where what had been thought to be one species, based on naked eye features, could be divided into two (or perhaps more) based on the evidence of the optical microscope. Thus, what had been one species was split into two or more. The advent of electron microscopes in the 20th century allowed the study of very fine morphological detail and cellular structure. In particular the structure of apothecia, perithecia and asci became important in classification of the ascomycetes in general. Some of those features had been used in ascomycete classification since the late 1800s but electron microscope studies gave more detailed structural information. Non-structural features have also been used and have also led to the lumping or splitting of species. LICHEN CHEMISTRY has been accepted as important for the identification of lichens from the late 1800s and has been used as a taxonomic tool for over a century, though there has been much heated debate as to the connection between CHEMISTRY AND TAXONOMY. The study of how thalli grow and develop their various features is another way of discerning similarities and differences and so have played an important role in classification schemes![]() .
.
Modern evolutionary theory developed from the later 1800s onward. Evolution results in change to organismal traits over many generations, leading to diversity in the descendants many generations hence. The division of organisms into species, genera and so on has always been a way of summarizing that diversity. The concept of evolution therefore provides a biological basis for a natural classification and it has become a goal to base the classification of living organisms on the evolutionary relationships between them. Ideally, to develop such a classification, the evolutionary history of lichens would be revealed by the study of numerous fossils of many ages which showed when various structural features appeared. Unfortunately the number of lichen fossils is not very large and there are few very old fossils. Hence, evolutionary relationships need to be determined, somehow, from what is found in lichens alive today. In the latter part of the 20th century there were various hypotheses as to how features such as micro-morphology, chemistry and thallus development indicated the degree of evolutionary closeness between species and such hypotheses were incorporated into classification schemes.
More recently there has been much emphasis on analysis of genomes. An organism's genome is its hereditary information, stored as DNA. Given that DNA is inherited, albeit mixed from both parents and with changes over time, makes genomic analyses attractive as a means of determining genetic and hence evolutionary closeness. In such analyses one looks at equivalent sections from the genomes of different species and the results are presented as sequences of letters, indicating how the four basic DNA components (adenine, cytosine, guanine, thymine) are arranged. If you have the DNA sequences from different species you can align them and then look for the number of differences between sequences. For example, suppose that the following are corresponding sections from the sequences of three species:
sequence 1: .....AGGTCATATTACCGAACTTA.....
sequence 2: .....AGTTCACATTCCCGAACTTA.....
sequence 3: .....AGTTGACATTCCCGAACTGA.....
In these parts the three sequences are very similar, with the three differences between sequence 1 and sequence 2 shown in red, and the two differences between sequence 2 and sequence 3 shown in blue. Essentially, counting the number of places in which sequences differ gives the basis for a numerical measure of closeness. You could say that, with regard to this section of DNA, species 1 is closer to species 2 than it is to species 3 since it takes three changes to transform sequence 1 into sequence 2, but five changes to transform it into sequence 3. This simplistic scenario is sufficient to explain the basic idea but in reality there may be many complications, fortunately irrelevant for the purpose of this page, in deriving measures of closeness between the three sequences.
All analysis (whether of growth form, spores, asci, secondary metabolites or genomes) is carried out on individual thalli. Various groupings of characteristics are used to define species, genera and so on, with fewer characteristics as you go up in taxonomic rank. Once you know the characteristics that define different species you can use the differences in the characteristics to derive some measure of similarity or, in an evolutionary classification, degree of evolutionary closeness.
Tree diagrams
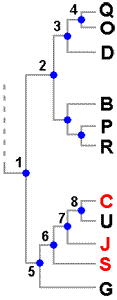 Once you have determined the degrees of closeness between different species you can display this information in a diagram such as the one alongside this paragraph, similar to a human family tree. The dotted line indicates that there are more species involved in the analysis, but we are interested in only the 11 shown here and indicated by letters.In this tree the blue dots, called nodes, are like ancestors in a family tree.
Once you have determined the degrees of closeness between different species you can display this information in a diagram such as the one alongside this paragraph, similar to a human family tree. The dotted line indicates that there are more species involved in the analysis, but we are interested in only the 11 shown here and indicated by letters.In this tree the blue dots, called nodes, are like ancestors in a family tree.
Since the classification that led to this tree is based on evolutionary principles and evolution involves changes between ancestors and descendents node 4 indicates a species ancestral to Q and O. Nodes 1, 2 and 3 are also ancestral to Q and O but 4 is the most recent common ancestor of Q and O while 3 indicates the most recent common ancestor of Q and D (and also of O and D). This diagram is saying that 3 marks the beginning of the divergence between the lineage that gave rise to D and the lineage that gave rise to the pair Q and O, while 4 marks a further point of divergence. Of the eleven species three have a trait called RED. This trait arose at some point and a diagram such as the one here can suggest hypotheses as to when that happened. One possibility is that 1, the most recent common ancestor of all eleven species had the trait RED. In that case the trait was lost by 2 (or some descendents of 2), by G and by U - in other words the trait was lost at least three times. If 1 had not been RED, then we could explain the current distribution of RED by positing a gain of RED by 6 and a loss by U - just two changes. This is a more economical hypothesis than the first one, but by no means proof that this in fact happened. As we are unable to analyse the ancestral species all we can say is that the current situation can be explained with a minimum of two changes in trait.
A tree such as this shows the species' evolutionary relationships, or phylogeny, but a phylogenetic tree is not itself a classification, though it can suggest possible classifications. In this example you might argue that there are three obvious genera here, namely the groups (QOD), (BPR) and (CUJSG). Someone else may argue that rather than three genera there should be just two, with the first two of the above three groups defined as the one genus. Each of those ideas is justified by the evidence presented by the diagram. A monophyletic group is defined as one consisting of an ancestor and all its descendants. Hence the group (3,4,Q,O,D), of which only the last three are extant today, is an example of a monophyletic group. If a classification is to be consistent with phylogeny then only taxa consistent with monophyly would be accepted. Hence (D,O,Q) would be an acceptable genus, as would be the other suggestions for genera just given. At an earlier level of knowledge the trait of RED may have been thought a significant feature and hence used to help define a genus consisting just of C, J and S. Current knowledge, as presented by the diagram, shows that RED is not a significant indicator of evolutionary closeness since C is closer to the non-RED U than to the other RED species. On the basis of the evidence presented by the diagram (C,J,S) is not monophyletic since the most recent common ancestor, 6, also has U as a descendant. The group (C,J,S) is paraphyletic - meaning that it does not include all descendents of the group's most recent common ancestor.
Despite the desirability of a monophyletic basis, paraphyletic groups are still widely used in lichen taxonomy. One factor is that much is still unknown. For example, if you wish to draw sound conclusions from genomic evidence it is important to study both the genomes of a large number of species and also different parts of each genome. Currently there are still many species for which little or no genomic information is available. Another point is that tree diagrams, which are very common in the lichenological literature, can look impressive but often there is some uncertainty in the evidence on which a tree is based or the one set of evidence is capable of generating two or more different trees. Trees that appear in publications typically have little numbers on the various branches. The production of trees is essentially a statistical process and the numbers are a measure of the unavoidable statistical uncertainty. You can look at those numbers as indicators of how well you could trust different parts of the tree and where more investigation is needed. Given facts such as these it is often a very sensible policy to keep using 'unsuitable' genera, until there is strong evidence in favour of a different classification. Moreover, there are different degrees of 'unsuitable' genera. In the artificial example given here, earlier evidence suggested that group C, J and S were closely related and the current evidence still supports a close relationship between the three so there is not a major change in the status of these three. The circumstances would be different if, for example, Q, B and C previously had been held to constitute a genus.
Now, a lichen tree
If lichenization had evolved just once and, once gained, had never been lost by descendents, all current lichens would form a monophyletic group and could then be treated as a self-contained group of organisms. Lichens do not form a monophyletic group. For a start, there are both ascomycete and basidiomycete lichenized fungi, albeit very few of the latter. Perhaps lichenization arose twice, once in the basidiomycetes, once in the ascomycetes and gave rise to two monophyletic groups - one of basidiolichens and one of ascolichens. The basidiolichens do not constitute a monophyletic group and neither do the ascolichens. The following figure shows where the lichenized ascomycetes fit into the ascomycetes as a whole and is based on genomic research published in 2009.
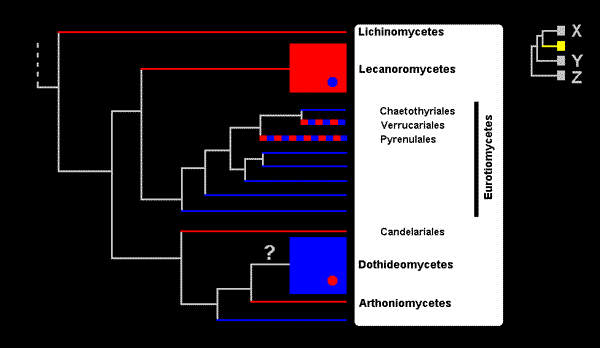
The figure is in two parts. The large tree on the left shows the classes that contain lichenized fungi with one class, Eurotiomycetes, given in more detail, the orders within that class being shown and three of them named. If a taxon consists only of lichenized fungi then it is shown as a red line. A taxon containing only non-lichenized fungi is shown by a blue line and taxa in which both lichenized and non-lichenized fungi are found are shown in red and blue. The lines for the latter are drawn thicker, simply to show the bicolouring more easily and the line thickness has no other significance. The class Lecanoromycetes consists almost totally of lichenized fungi but there is a small number of non-lichenized species within the class, hence the blue dot within the Lecanoromyctes. This class contains the majority of the lichenized fungi (containing about 14,000 of the approximately 18,000 known lichen species). The Dothideomycetes is the largest class within the ascomycetes and contains about 19,000 species - but only a small number of lichenized species. The Verrucariales and Pyrenulales consist mostly of lichenized fungi. The yellow area in the small tree to the right shows where the large tree fits into the tree of all ascomycetes. As you can see there are three other groups of ascomycetes, all non-lichenized, and they have been labelled X, Y and Z. The first of those groups contains about 18,000 species while Y and Z, combined, contain about 3,000.
At the extreme left of the large tree is the most recent common ancestor of all the taxa in that tree. Either that ancestor was capable of forming a lichenized association or it wasn't. In either case the current distribution of lichenized and non-lichenized taxa calls for one or more losses or gains of lichenization to account for the current mix of lichenized and non-lichenized taxa. Here are some of the hypotheses supported by the evidence behind the above tree.
There was a single lichenization in the immediate ancestor of the Lecanoromyctes and at least one loss of lichenization within that class. The immediate ancestors of each of the Lichinomycetes, Candelariales and Arthoniomycetes was lichenized. The Dothideomycetes is the one class within which lichenization has occurred several times independently but the evidence is insufficient to determine whether the immediate ancestor of the Dothideomycetes was lichenized or not - hence the question mark. With regard to the group consisting of the Chaetothyriales, Verrucariales and Pyrenulales the evidence is unclear. It does not distinguish between the hypothesis of two gains of lichenization (early in each of the Verrucariales and Pyrenulales) from a non-lichenized common ancestor and the hypothesis of a gain of lichenization by a common ancestor of the Verrucariales and the Pyrenulales, followed by a loss of lichenization in the Chaetothyriales. Regardless of that, the evidence supports the hypothesis that there has been at least one loss of lichenization within each of the Verrucariales and the Pyrenulales.
As more taxa and larger sections of genomes are studied some conclusions may change. This is shown already by the fact that the more extensive investigation behind the 2009 publication did not support an earlier hypothesis, published in 2001, that lichenization was the ancestral state for the majority of current ascomycete taxa. However, while there are differences between the conclusions of 2001 and 2009, both studies supported the hypothesis that lichenization had been gained and lost several times![]() .
.
Apart and together
Lichens are classified by the fungal partner, so all lichens belong to the fungal kingdom. Though this fungal-based taxonomy of lichens has been accepted for over a century, for much of that time lichenized and non-lichenized fungi have been studied largely as separate subjects. As one lichenologist wrote in 1973:
Even a superficial analysis of any of the past systems will show that we cannot really speak of a true integration of lichenized fungi in any presently accepted fungal system. Lichen systematists have hardly ever been really familiar with the corresponding fungal groups, and mycologists have had enough difficulties with their own groups without bringing in the lichenized fungi
.
Twenty-one years earlier another lichenologist had written:
Whereas in the last half-century the classification of Ascomycetes - the fungal class of overwhelming importance in the present study - has been completely reformed, the lichen system presented by Zahlbruckner in 1903-07 is still accepted and used by most lichenologists. This is due not so much to any excellence inherent in it as to the lack of interest manifested by most lichenologists vis-à-vis taxa above species level, and to their lack of knowledge of the general taxonomy of fungi
.
The person referred to was Alexander Zahlbruckner (1860-1938), an Austrian lichenologist, and in relation to Zahlbruckner's classification the same author noted that "...it has become dominating in a way strongly detrimental to the development of a more natural lichen classification".
It would be natural to ask how the situation described by these quotations could be tolerated for so long. Has it not been harmful to a proper classification of the lichenized fungi - and also of the non-lichenized fungi? It is worth repeating some numbers given earlier. About 64,000 ascomycete species are known, of which about 18,000 are lichenized. The Lecanoromycetes account for about 14,000 of the known lichenized ascomycetes and the second largest lichenized class, the Arthoniomycetes, for about 1,500. A very large proportion of lichens belong to just one class and, on current evidence, that class arose from one gain of lichenization. It is therefore possible to do much work on lichen classification by working entirely within that class, in isolation from the remaining ascomcyetes. Certainly if you are interested in the origins of that class, you need to think of ascomycetes as a whole, but once you are studying the species within that class the rest of the ascomycetes are largely irrelevant. The species within the Arthoniales form a similar self-contained group. This is not a denial of the need to look at fungi as a whole. Instead, the small number of gains or losses of lichenization has meant that it has been possible to study large groups of ascomcyetes more or less in isolation from each other. The situation would have been quite different if the lichenized fungi were spread widely through the fungal kingdom. This would have produced many additional cases like the Pyrenulales and the Verrucariales, with more intermingling of lichenized and non-lichenized fungi. In such a situation a non-integrated approach would have been worthless from the beginning. Another fact is that over the past twenty years there has been much new evidence relevant to fungal phylogeny as a whole, considerable changes in classificatory ideas and during those twenty years there has been integration of lichenized and non-lichenized fungi.
The period before then was not wasted. There was enough productive work to do in accumulating the essential species information that has allowed the better understanding of fungal (and hence also lichen) phylogeny in recent times.
![An Australian Government Initiative [logo]](/images/austgovt_brown_90px.gif)